You’d find valence electrons at the outermost shell of an atom. Lewis structure helps us determine valence electrons and how it helps any chemical reaction and predict the type of bond. Argon has ten core electrons present in shells 1 and 2.Ĭalculate valence and core electrons of Carbon, Silicon, Nitrogen, Phosphorus, Oxygen, Sulfur, Magnesium and Calcium.What is a valence electron? Valence electrons are the electrons in the outermost shell of an atom, while the electrons which are mostly present within the inner circle are known as core electrons.
The outermost shell is 3, and the number of valence electrons is 8 (3s 2 3p 6). Argon has electrons distributed among three shells (1, 2, and 3). The electronic configuration of Argon is 1s 2 2s 2 2p 6 3s 2 3p 6. The 1s 2 electrons are the core electrons of the Carbon atom.Īn Argon atom has the atomic number 18. Therefore, the number of valence electrons of Carbon is 4 (2s 2 and 2p x 1 2p y 1). Since the valence electrons are the outermost shell electrons, here it is the electrons of shell 2. The 1 and 2 are two different shells of the Carbon atom. The electronic configuration of carbon is 1s 2 2s 2 2p x 1 2p y 1. The outermost shell containing the valence electrons is determined from the electronic configuration of the element.Ī Carbon atom has an atomic number of 6. The electrons are distributed in different energy levels, and their representation is known as the electronic configuration of the element. The atomic number represents the number of protons or electrons in an atom.
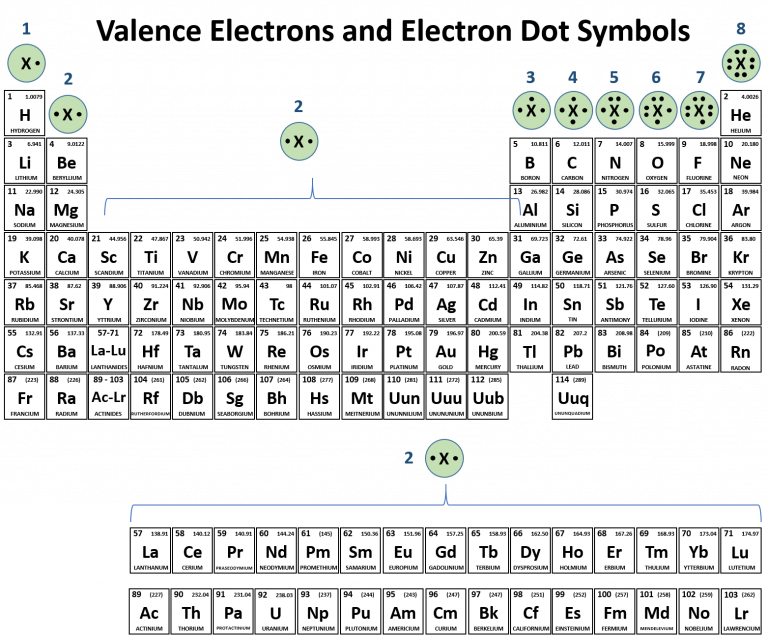
Therefore, the valence electron shell of Fluorine is 2 (2s 2 2p 5), whereas for Chlorine, it is 3 (3s 2 3p 5), and Bromine is 4 (4s 2 4p 5). However, the size of the halogen increases down the group, and new electrons are added in new shells. The only difference will be their shell number due to the increase in atomic size.įor example, all halogens belong to group 17 of the periodic table. The group number (column number) represents the number of valence electrons common to all the elements within that group.Īll elements belonging to the same group in the periodic table will have the same number of valence electrons ( table A ). This lowers their energy and contributes to stability, making them indifferent to bonding reactions. Their attractive interactions with the nucleus are stronger therefore, they are bound tightly to the nucleus. The core electrons are those present closer to the nucleus. Some remain uninvolved as lone pair(s) or nonbonding electron(s). However, not all valence electrons participate in bond formation or other chemical reactions. Most Lewis structures use valence electrons as dots above the atom’s symbol for structural representation and in chemical reactions. The loss or gain of the valence electrons forms ions that electrostatically attract to form ionic bonds.


Consequently, the valence electrons are loosely held therefore, they participate in chemical reactions by being gained, lost, or shared.įor instance, two atoms share any one valence electron to form a two-electron covalent bond. Valence electrons are the outermost, farthest from the nucleus, and the nuclear attraction.
